See feature articles below:
About Broad Street Alerts:
Big opportunities in Small Cap’s
Broad Street Alerts recent profiles and track record, 153% in verifiable potential gains for our members on the last 3 small cap alerts alone!
February 10th, 2016- (NASDAQ: BONT) opened $1.65/share hit a high of $3.00/share within 30 days our member potential gains- 83%
March 7th, 2016-(NYSE-MKT: FSI) opened at .91/share and hit 1.10/share within 5 days for gains of 21% for our members.
March 24th, 2016- (NASDAQ: ICLD) opened at $.77/share it a high of $1.15/share within 2 days for gains of 49% for our members.
These are numbers that make traders drool. Any trader in any market would fall all over themselves to see numbers like this. So if you’ve been on the fence, perhaps it’s time to start doing some research and verify our numbers for yourself. We are constantly raising the bar and separate ourselves from the rest of the small-cap newsletters as the best in business.
We know with a large following comes a large responsibility as we have everyone from institutional investors to the beginner following our profiled securities in our newsletters. This is something we take very seriously always seeking small cap growth companies that have both near and long-term potential for our members.
***Get our small cap profiles, special situation and watch alerts in real time. We are now offering our VIP SMS/text alert service for free, simply text the word “Alerts” to the phone number 25827 from your cell phone.
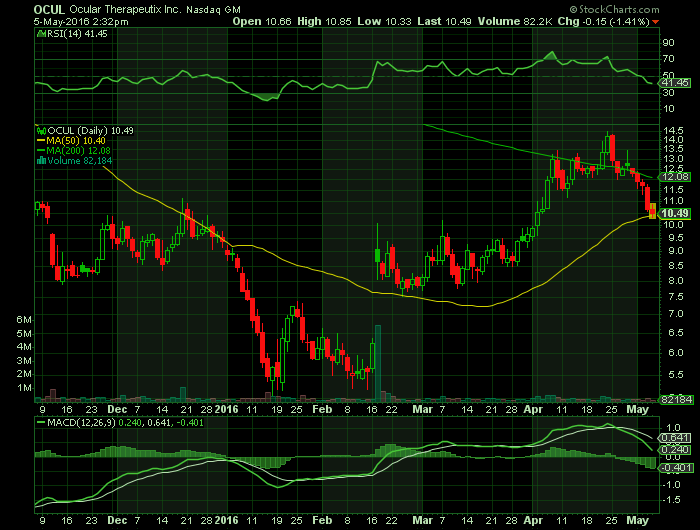
Report on: OCUL
Ocular Therapeutix, Inc. (OCUL), a biopharmaceutical company focused on the development and commercialization of innovative therapies for diseases and conditions of the eye, today announced that it has completed its End-of-Phase 2 review with the U.S. Food and Drug Administration (FDA) for its OTX-TP (sustained release travoprost) product candidate for the treatment of glaucoma and ocular hypertension.
Based on this review, the company intends to initiate the first of two Phase 3 clinical trials during the third quarter of 2016. The FDA has stated that it agrees with the overall Phase 3 clinical development program proposed by Ocular Therapeutix in its briefing package submitted to the FDA, which includes the following:
Two Phase 3 clinical trials that will include an OTX-TP treatment arm and a placebo-controlled comparator arm that would use a non-drug eluting hydrogel-based intracanalicular depot.
The Phase 3 study design will not include a timolol comparator or validation arm.
The study design will not have eye drops, placebo or active, administered in either arm.
A primary efficacy endpoint of statistically superior and clinically meaningful reduction of intraocular pressure (IOP) from baseline with OTX-TP compared to placebo at 60 and 90 days. The FDA has also indicated that they will take into consideration the risk-reward profile of OTX-TP relative to currently available therapies in their evaluation of the Phase 3 clinical trial results.
“We are pleased with the End-of-Phase 2 feedback from FDA concerning our Phase 3 clinical development plan, which proposes a trial design that compares OTX-TP with a placebo arm. We believe this reflects a real-world appropriate clinical study design for this drug product candidate,” said Amar Sawhney, Ph.D., President, Chief Executive Officer and Chairman. “OTX-TP has shown a clinically meaningful IOP-lowering effect in clinical trials to date, and may offer an important advancement in the treatment of glaucoma. Importantly, we believe OTX-TP may be able to address the major issue of low patient compliance rates associated with currently approved topical therapies and their chronic, burdensome dosing schedules while potentially improving the safety profile due to the absence of preservatives.”
About Glaucoma and Ocular Hypertension
Glaucoma is a chronic, sight-threatening diseases in which elevated levels of intraocular pressure are associated with damage to the optic nerve, which may result in irreversible vision loss. Glaucoma is the second leading cause of blindness in the world. Ocular hypertension is characterized by elevated levels of intraocular pressure without any optic nerve damage. Patients with ocular hypertension are at high risk of developing glaucoma. In the U.S. alone 2.7 million people suffer from glaucoma. According to IMS Health data, there were 34 million prescriptions and sales of over $2.7 billion of drugs administered by eye drops for the treatment of glaucoma in the U.S. in 2015.
Compliance is seen as the biggest problem with existing therapies for glaucoma, and more than 50% of patients on topical prostaglandin analogs are not compliant with their therapy within the first six months of treatment.
More About OTX-TP (Sustained Release Travoprost)
OTX-TP (sustained release travoprost) is a preservative-free drug product candidate that resides within the canaliculus and delivers the prostaglandin analog travoprost to the ocular surface for up to 90 days. The drug depot is designed to deliver a continuous steady release throughout the treatment period. A fluorescent visualization aid is formulated within the product to enable both the physician and the patient to monitor drug presence throughout the course of therapy.
About Ocular Therapeutix, Inc.
Ocular Therapeutix, Inc. (OCUL) is a biopharmaceutical company focused on the development and commercialization of innovative therapies for diseases and conditions of the eye using its proprietary hydrogel platform technology. Ocular Therapeutix’s lead product candidate, DEXTENZA™ (sustained release dexamethasone) Intracanalicular Depot, is in Phase 3 clinical development for post-surgical ocular inflammation and pain and allergic conjunctivitis, and in Phase 2 clinical development for dry eye disease. A New Drug Application (NDA) for the post-operative ocular pain indication has been filed with FDA and has a Prescription Drug User Fee Act (PDUFA) date of July 24, 2016. A third Phase 3 clinical trial is being conducted for post-operative ocular inflammation and pain. For glaucoma and ocular hypertension, a Phase 2b clinical trial has been completed with OTX-TP (sustained release travoprost) and the first of two planned OTX-TP Phase 3 clinical trials is expected to be initiated in the third quarter of 2016. Ocular Therapeutix is also evaluating sustained-release injectable anti-VEGF drug depots for back-of-the-eye diseases. Ocular Therapeutix’s first product, ReSure® Sealant, is FDA-approved to seal corneal incisions following cataract surgery.
Source – Company Press Release
Broad street alerts has not been compensated for the mention of any publicly traded companies in this article nor do we own positions in any of the companies in this article.
Stock market
Hot small cap stocks
small cap stock picks
Biotech stocks
FDA approval stocks
FDA calendar
Trade stocks
Become a day trader
Day trade stocks for a living
PDUFA date set
micro cap stocks
Best stocks 2016
Hottest small cap stocks
Best stock picks
Who to follow for stock picks
Apple news stock picks
Stock picks on apple news